Experiment of The Month
Non-Coalescing Coffee Drops
The forces that hold a falling drop of water in a spherical shape can be strong enough that the drop fails to merge immediately with another drop of water. The effect can be even seen when a falling drop meets the top surface of a pool of water.
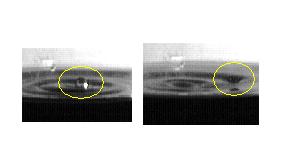
While keeping vigil at his coffee pot, Dr. Cooney noticed that this "non-coalescence" was remarkably visible for drops rebounding from the coffee dripping from above into the half-full pot. Senior Arzie Yeater took up the challenge of making careful observations and of understanding the phenomenon. The figure at the below shows two frames of his high-speed video for one non-coalescing drop, in motion from left to right.

Mr. Yeater used our high speed (500 frames per second) camera to follow the evolution of non-coalescing drops. He also refined the experiment, manufacturing his own drops rather than using a coffee pot. Interestingly, however, he still needed a weak coffee solution to get the drops to form reliably.
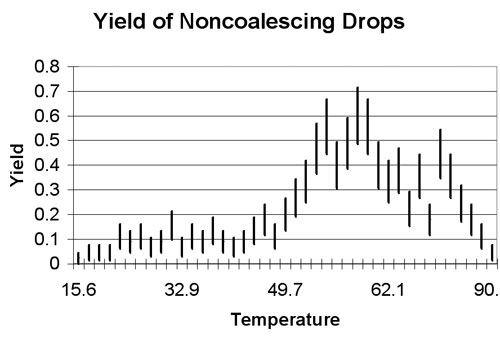
In the picture (below), he drops coffee from a straw (used like a pipette) while recording with the camera seen in the lower part of the photo. The adjustable hotplate maintained a constant temperature for the water. Mr. Yeater compiled data for the quality of non-coalescing drops as a function of the temperature of the water. He found that the optimum temperature was about 50C.

He measured quality by the "yield," the fraction of drops from the straw that resulted in a non-coalescing drop, formed on the rebound. The figure shows how the non-coalescing drop is formed.
If the water is deep enough, the falling heavy drop does not produce the famous "crown." Instead, it produces a column of water shooting up at the center of the fall location. When that column falls down, its top often pinches off, leaving a small drop to fall by itself. Drops like this are the non-coalescing drops.
The figure shows the yield that Mr. Yeater measured, plotted versus water temperature. The graph at the right clearly shows a peak between 50C and 65C. A yield of .5 means that half the time he released a drop, he created a non-coalescing drop.
He has proposed two mechanisms that keep the drop from merging with the pool of water below it: A pressure gradient from water evaporating up from the pool, and surface tension which makes it energetically favorable for a small drop to remain intact.
Surface tension increases at lower temperatures, and the evaporation increases at higher temperatures. In this model, drops form most favorably when both effects join together.
Mr. Yeater's movies are online and may be downloaded from the list below.
- Double
- Short
- Shorter
- Even Shorter
- Good One
- LightCoffee
- Fuzzy
-
Contact Information
Contact Number: 717-871-4297
Email: physics@millersville.edu